|
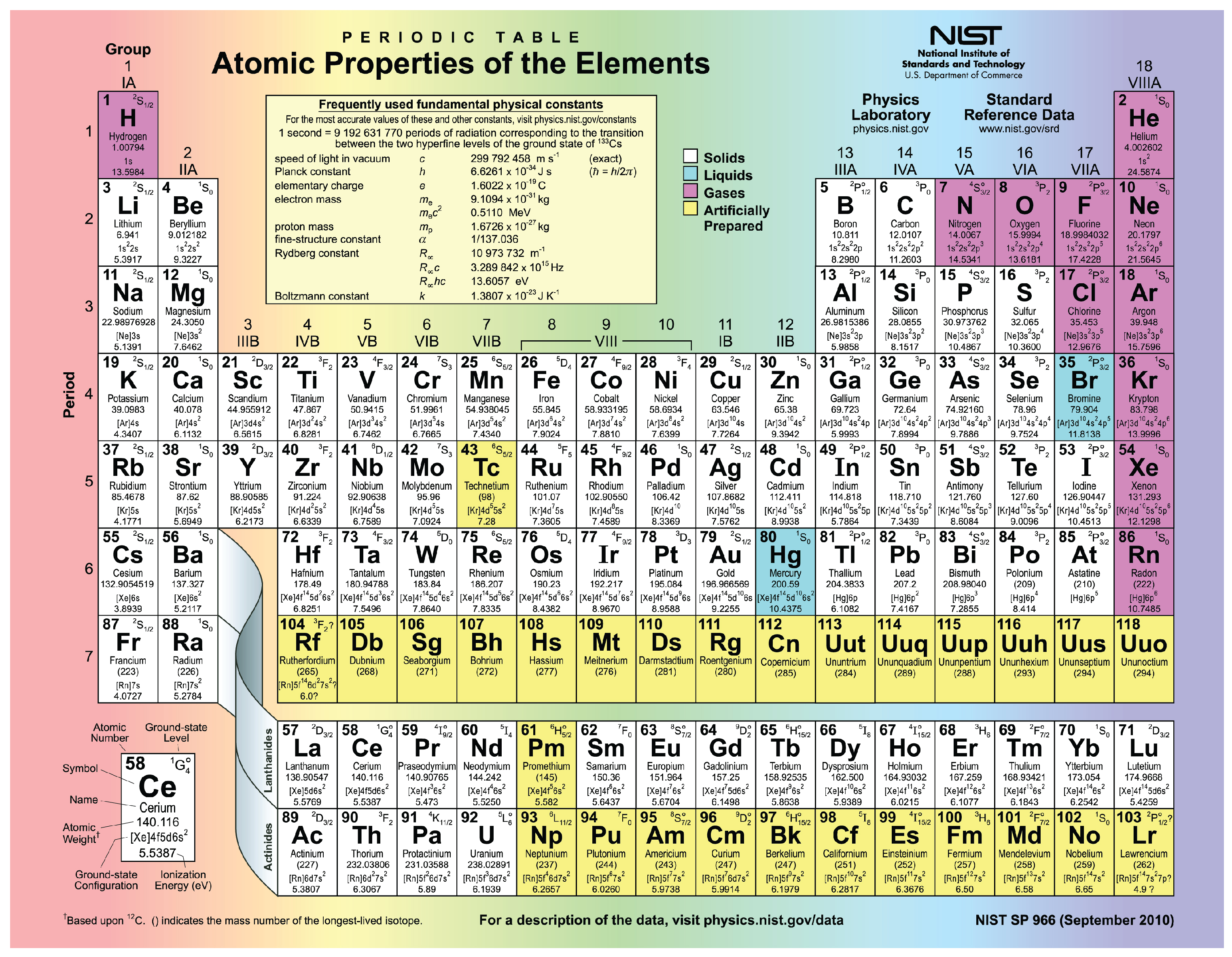
According to Mendeleev’s Periodic Law, “ Physical and chemical properties of elements are periodic function of their atomic masses”.This happens as the tendency of an atom to gain electrons increases due to increase in nuclear charge, the valence electrons are pulled in strongly by the nucleus and it becomes easier for an atom to gain electrons. This happens as the tendency of an atom to lose electrons decreases due to gradual increase in the number of protons and nuclear charge.ĩF has the highest nonmetallic character because non-metallic character of an elements increases as we move from left to right in a period of the periodic table. Due to large positive charge on the nucleus, the electrons are pulled closer to the nucleus and the size of atom decreases.ġ1Na has the highest metallic character because metallic character of an elements decreases as we move from left to right in a modern periodic table. According to the trend, as we move from left to right atomic size of an atoms decreases. Due to large positive charge on the nucleus, the electrons are pulled closer to the nucleus and the size of atom decreases.ĤBe has smallest atomic radius because 19K, 3Li, 11Na are present in same group 1 but Be is present in group 2. The atomic number of elements increases which means the number of protons and electrons in the atoms increases. This happens as the tendency of an atom to lose electrons increases due to decrease in nuclear charge and increase in numbers of shell.ġ1Na has largest size among these because according to the trend, atomic radius decreases as we move from left to right in a period of the periodic table. According to the trend, electropositive character of an elements increases as we move from top to bottom in a group of the periodic table. K,L,M.Ģ0Ca, 4Be belong to second group because these elements have 2 electrons in its outermost shell.ĨO is the most electronegative element among these because electronegativity increases as we move from left to right in a period of the periodic table.ġ3Al is the most electropositive element among these because 4Be, 6C, 8O, 5B belong to same period, but 13Al belong to next period. The coefficient multiplies that by #4#, so we get #8xx4=32# atoms of hydrogen.Electronic configuration of the following elements is:ġ4He, 11Na, 15P belong to the third period because according to their electronic configuration, each element contains three shell i.e. #H:# Ignoring the coefficient, we know there's #5+3=8# atoms of hydrogen. #C#: It only appears once and without a subscript, so #1xx4=4#. Again, counting the coefficient, there's #4xx2=8# atoms of nitrogen. #N#: The bracket around #NH_2# has a subscript of #2#, so that means there's #2# nitrogen atoms if we're not counting the coefficient. #H#: There's #3+1=4# atoms of hydrogen without the coefficient (it appears once as #H_3# and once as #H#), so there's #4xx5=20# atoms of hydrogen with the coefficient. After looking at the coefficient, though, there's #2xx5=10# atoms of carbon. That means that there are #2# carbon atoms if we don't look at the coefficient. In both cases, they don't have subscripts. #C#: Carbon appears twice in this formula. 
It also means that there's #2xx2=4# atoms of oxygen. When we put a coefficient in front of something, we're multiplying the entire thing by that number.įor example, in #2CO_2#, there are #2#CO_2# molecules.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
